Introduction Supply chain digitalization represents a fundamental shift in how organizations operate, moving beyond simply digitizing paper-based processes. While many business leaders use the terms interchangeably, supply chain digitization refers to converting analog information into digital formats. In contrast, supply chain digitalization involves transforming operations using digital technologies to make them smarter, faster, and more integrated. In this […]
Introduction EDI reporting refers to generating detailed reports and dashboards from EDI transaction data. These reports can track important metrics like transaction volume, order fulfillment accuracy, inventory levels, on-time delivery, and more. By analyzing this information, businesses can optimize operations, reduce errors, and improve collaboration with suppliers. For example, supply chain managers can set up […]
Introduction EDI support has revolutionized supply chain management, driving efficiency, accuracy, and speed across industries. When businesses implement real-time data exchange through EDI, key players in the supply chain receive prompt updates about inventory levels, shipping schedules, and payment status. What makes EDI in supply chain management so powerful? For starters, these automated transactions can […]
Introduction The GS1 Global Data Model (GDM) represents a revolutionary approach to standardizing product information exchange across global supply chains. This comprehensive framework harmonizes product attributes, creates unified data standards, and enables seamless communication between trading partners worldwide. By implementing harmonized product attributes through the GS1 Global Data Model, businesses can achieve unprecedented levels of operational efficiency, […]
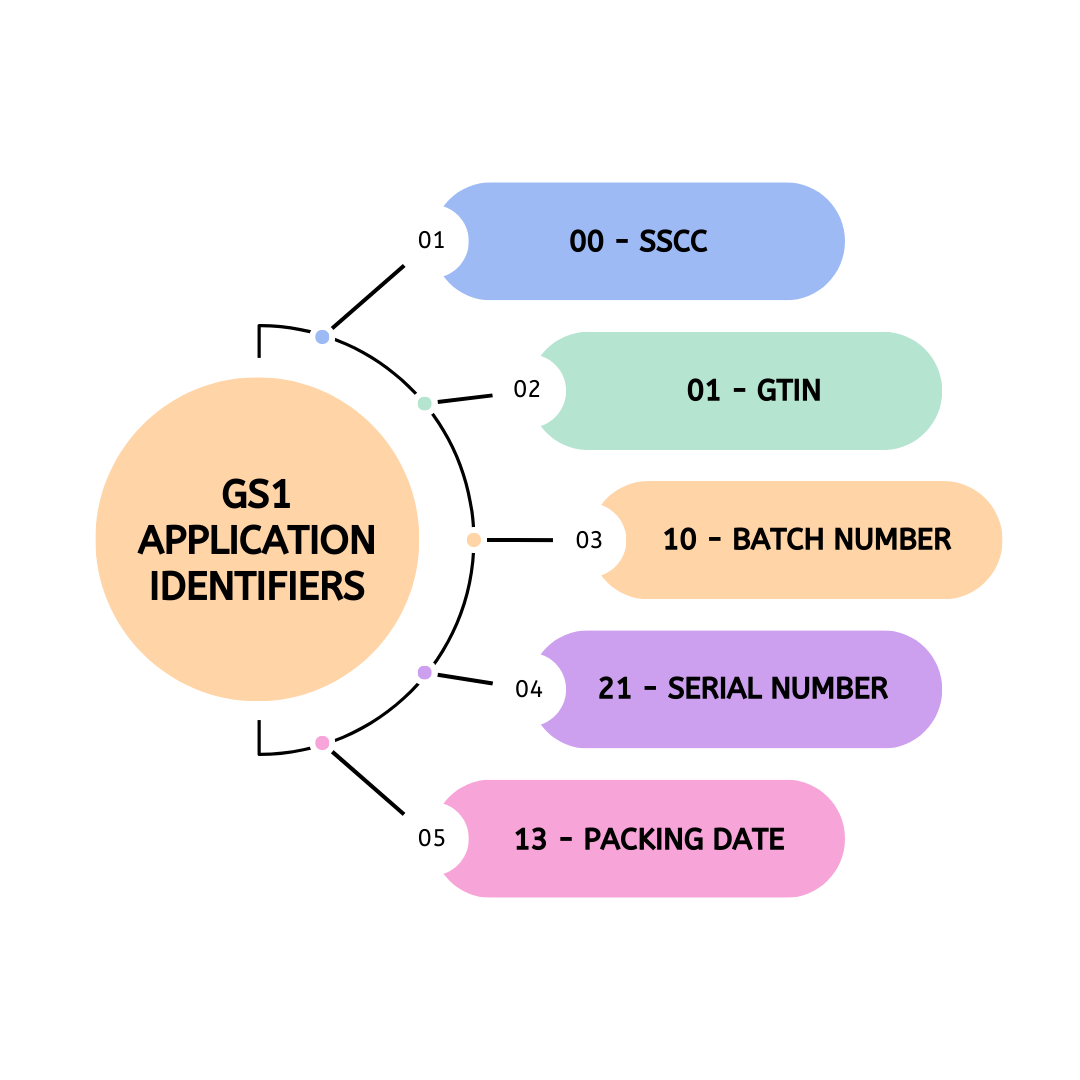
Introduction GS1 Application Identifiers serve as the essential language of modern barcodes, translating coded information into meaningful data for supply chain operations. These identifiers consist of two, three, or four-digit prefixes that define both the meaning and format of the data that follows them. Essentially, they give context to what would otherwise be just a string of […]